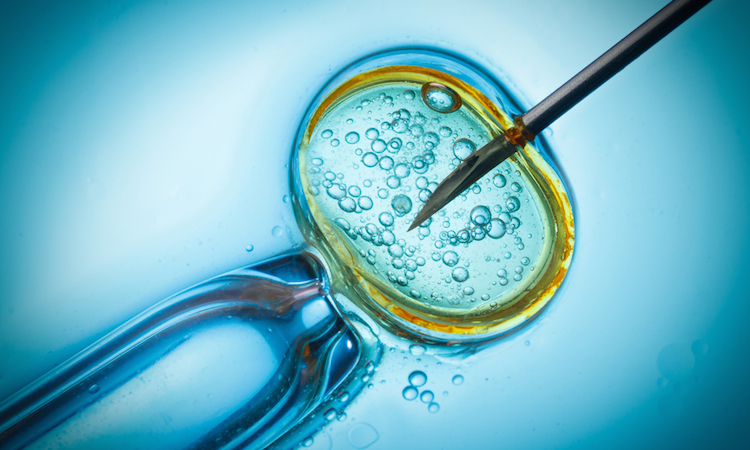
Although experimental and applicable to a small population of individuals, mitochondrial DNA replacement therapy is a procedure that holds a number of exciting possibilities. The method has attracted its share of controversy, largely because it involves combining genetic material from two different women’s egg cells.
In recent months, however, a new report from the UK has said that this proposed In vitro fertilization method is likely safe. However, the Federal Drug Administration (FDA) in the US and the Human Fertilisation and Embryology Authority (HFEA) in the UK are both adamant that further tests and research are needed before this therapy is used in patients.
As may remember from your high school or college biology classes, the mitochondria are organelles that produce energy at the cellular level. They often get referred to as the power plants of our cells. These special organelles have their own DNA, called mtDNA, which can carry mutations that cause a host of diseases.
Major organs like the brain and heart are sometimes affected by mitochondrial diseases, as well as major muscle groups and more. Because mitochondrial DNA is inherited from the mother, scientists are now using this technique to transplant a healthy donor’s mtDNA to embryos, allowing mothers to have healthy children free of mitochondrial disease.
As things stand currently, mitochondrial DNA replacement therapy is not legal in the UK and the FDA has said that, although not technically illegal, the procedure needs to have to FDA’s stamp of approval before it can be performed in the United States.
Dr. Jeffery Fisch of Green Valley Fertility Partners in Las Vegas, Nevada agrees that if additional studies answer some of the outstanding questions, the time would be right to begin clinical trials in women who have very high risk of passing a severe mitochondrial disease to their children.


No Comment